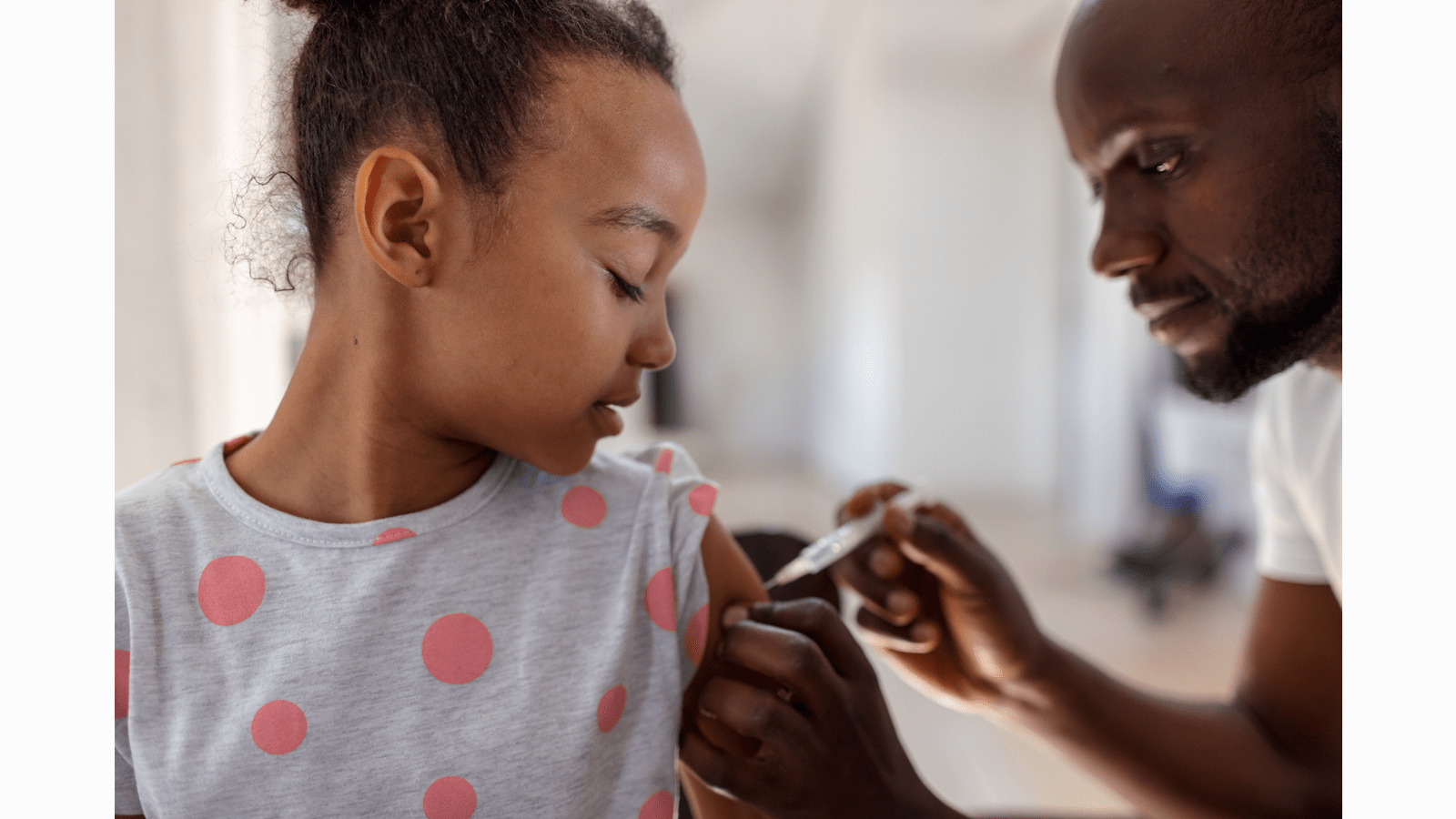
What’s it like raising twin boys? Danielle, a primary immunodeficiency patient, shares the journey of her first year as a mom. #WorldPIWeek https://bit.ly/3UsiFXN
World PI Week: 22 -28 April
In honor of this week, meet Danielle and learn how she is managing motherhood (of twins!) and a Primary Immunodeficiency.
CSL has Announced its 2024 Half Year Financial Results. Click here to read more →
CSL. Always Evolving for a Better World.
CSL has delivered biotechnology excellence for over a century. Today, with the combined expertise of CSL Behring, CSL Plasma, CSL Seqirus and CSL Vifor, CSL’s offerings are more diverse than ever to help ensure patients and people everywhere get the treatments they need. We’re always improving so life can, too.
Our Businesses and Products
CSL. Always Evolving for a Better World.
CSL has delivered biotechnology excellence for over a century. Today, with the combined expertise of CSL Behring, CSL Plasma, CSL Seqirus and CSL Vifor, CSL’s offerings are more diverse than ever to help ensure patients and people everywhere get the treatments they need. We’re always improving so life can, too.
Our Businesses and Products
CSL Behring. Leading the Way in Treating Rare and Serious Diseases.
CSL Behring is a global biotech leader with a broad range of biotherapies for rare and serious diseases including bleeding disorders, immunodeficiencies, hereditary angioedema, neurological disorders and Alpha 1 Antitrypsin Deficiency.
More About CSL Behring
CSL Seqirus. On the Front Line of Public Health Protection.
As the only vaccine company with a singular global focus on influenza, we work tirelessly on cutting-edge research, transformative technologies, and the latest methods of production and distribution. Together with our partners we’re on the front line every day, working to help protect communities from seasonal influenza and global pandemic threats.
More About CSL Seqirus
CSL Vifor. Changing the Game in Iron Deficiency and Nephrology.
The newest member of the CSL family, CSL Vifor is a global leader in iron deficiency and nephrology and is committed to launching the next generation of therapies to truly address the full spectrum of kidney disease, with a focus on dialysis and rare disease.
More About CSL ViforVita: Original Stories
'Vita' means life. It's also where we share the stories of biotech's promise to patients and public health.

Careers at CSL
We are committed to helping employees fulfill their career aspirations while working in a values-based culture. Are you next?